
Knowing how numbers work and how ratios work is KEY to understanding and working chemistry stoichiometry problems. You'll only have to go to grams IF the number of moles is asked for.
Just work the problem in pounds - it will work.

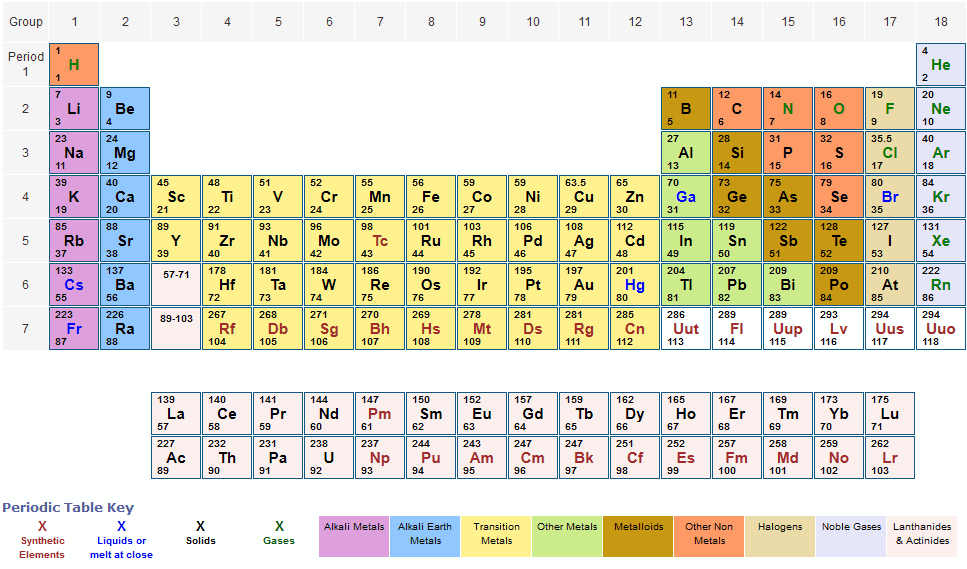
there is really no reason to convert to grams first and then back out to pounds later. How? Well IF the problem is stated in say pounds, and then wants the answer in pounds. Quite the rigorous path for "all" problems. And finally, convert those grams into any other unit needed that might be asked for. Then (if need be) convert your answer in moles into grams. Then, convert those grams in to moles and work the problem in moles only. In general, to work all types of stoichiometry problems, we say to convert all masses to grams first. Those atomic weights are the number of grams you will need of that element in order to have exactly 1 mole of that element. Counting by number is the molar amount, while measuring by mass is the. This helps tremendously when having to convert from moles to mass as we often do in chemistry. Because of that old definition, we were able to say that all those atomic weights are in grams per mole of substance or abbreviated g/mol. So why DO we seem to concentrate on the "gram" as our go to guy on the periodic table for atomic weights and ultimately for molar masses and molecular weights? Well the key here is the way we historically defined the mole. All chemical ratios work just as well with masses as they do with our oh so familiar moles. You can work chemistry mass problems in any mass you want and it will still work because the masses are relative to each other. Not to mention the myriad of masses represented by all the metric prefixes to prepend to "gram". short tons, long tons, drams, grains, or stones. You could think in pounds, or kilograms, or ounces, or even tons, or heaven forbid. Relative masses means that they are all corrected relative to each other. BUT it would be much much better for you to realize that those could be ANY unit of weight/mass you choose and the whole table would still be correct. "Well, I know the weights are in grams because that is how I learned it in high school". Notice how the atomic weights have no units after them. Hey you! LOOK again at any periodic table - including the one above. The diagram below illustrates the parts and their definitions. You need to make sure that you know what each of these parts is and what it represents.
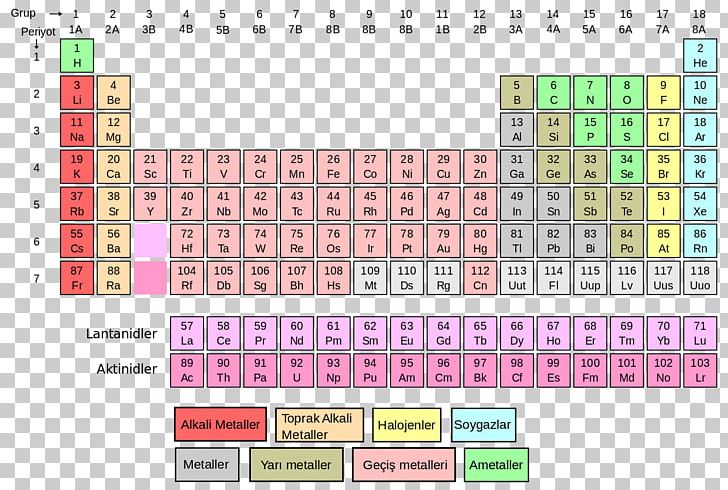
You want a lot more periodic tables to chose from? *Note: If you click on the table, you'll launch it into its own window/page on your browser. These three pieces of data are the elemental symbol, the atomic number (typically given the symbol, Z, and the atomic weight. In it's simplest form (shown below), each entry only has three pieces of information that you will need to know. Step 6: Calculate - Using the given atomic masses and the number of atoms in one water molecule, the calculated molar mass of H₂O is approximately 18.016 g/mol.The periodic table can often be presented with an abundance of data about each and every element listed. Step 4: Determine the Number of Oxygen Atoms - There is one oxygen atom in one water molecule. Step 3: Determine the Number of Hydrogen Atoms - There are two hydrogen atoms in one molecule of water. Step 2: Identify the Atomic Mass of Oxygen (O) -The atomic mass of oxygen is approximately 16.00 grams/mol. Step 1: Identify the Atomic Mass of Hydrogen (H) - The atomic mass of hydrogen is approximately 1.008 grams/mol. Molar Mass (H₂O) = Atomic Mass of H × Number of H atoms + Atomic Mass of O × Number of O atoms Steps to determine the molar mass of H 2O: For water (H₂O), which consists of hydrogen (H) and oxygen (O), calculating the molar mass involves summing the atomic masses of its constituent atoms. The molar mass of a chemical compound represents the mass of one mole of that substance.
